In-use stability studies are typically conducted in the later stages of drug development – once the DP formulation has been finalized and just before entering clinical trials and the regulatory approval phase.
However, this can be very risky as the recovery rates of infusion devices and the adsorption phenomena on contact materials (syringes, CSTDs, infusion lines, etc.) during use (“in-use”) are still unknown.
At ProJect Pharmaceutics we preventatively integrate in-use stability trials early informulation development, helping our customers make data driven decisions for their formulation lock.
We are pleased to present a confirming example in the following case study:
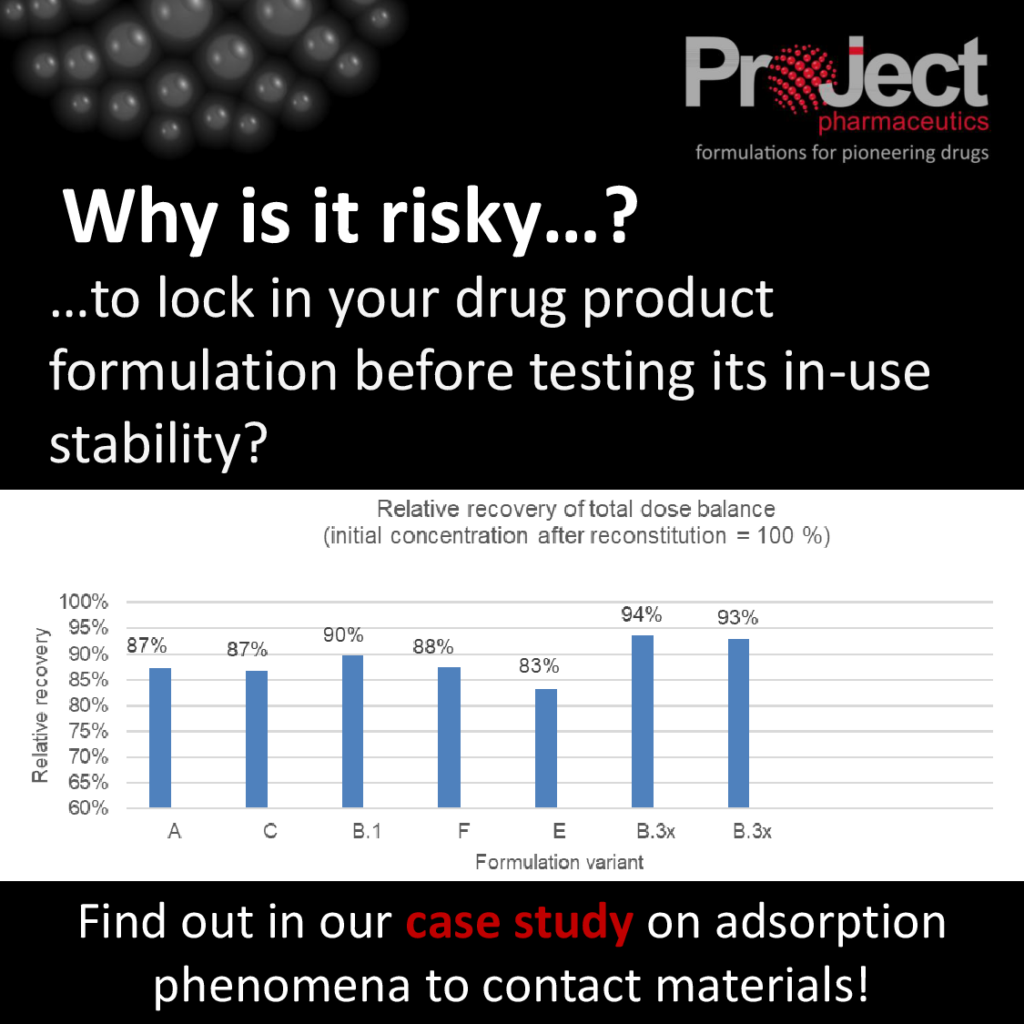
- The 5 most promising formulation candidates were investigated in a simulated infusion procedure
- A difference of up to 10 % recovery were observed between formulations
- Formulation B.3x was able to significantly reduce the adsorption phenomena even further
The formulation composition of B.3x was derived by back-calculating data from in-use stability studies on B.1. It was then optimized to determine “how much content of a particular inactive ingredient is required to prevent the observed adsorption phenomena”. Our customer was thus in the lucky position to select their final formulation based on in-use stability data generated during drug product development, leading to a more robust and reliable drug product entering into clinical studies.
ProJect Pharmaceutics CRO is an independent, European development partner offering in-use stability studies for injectable drug products from early formulation development to late stages just ahead of clinical trials. We test the DP compatibility with i.e. common infusion media as well as infusion and injection equipment early during formulation development.